Patented Molecule Request
Request access to Prof. Durdağı's patented therapeutic compound
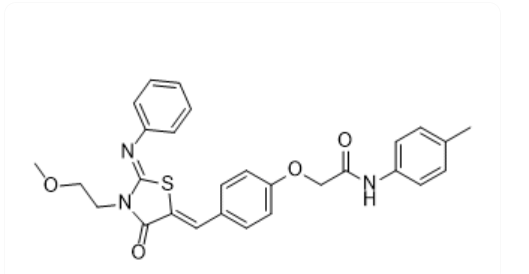
BAU-243 Molecule Structure
Novel BH3 mimetic Bcl-2 inhibitor | Patent by Prof. Serdar Durdağı
⚠️ NOT FOR HUMAN USE - LABORATORY USE ONLY
Compound Identifiers
BAU-243 (BAU243, AN-698/40780701)
Physicochemical Properties
- Molecular Weight: 501.60
- Formula: C₂₈H₂₇N₃O₄S
- Solubility: 10 mM in DMSO
Publication
View Nature ArticleAvailable Quantities
- 1 milligram
- 5 milligrams
- 10 milligrams
Submit Request
Fill out the form to request this molecule for research purposes
About BAU-243
BAU-243 is a novel BH3 mimetic Bcl-2 inhibitor with high binding affinity that effectively reduces glioblastoma multiforme (GBM) cell proliferation, including cancer-initiating stem cell subpopulations. This compound represents a breakthrough in targeting tumor-initiating cells that are often resistant to conventional therapies.
Key Mechanisms
- • Disrupts Beclin 1 and Bcl-2 interaction to activate autophagy
- • Induces autophagic cell death regardless of Bcl-2 expression levels
- • Promotes cell cycle arrest in low Bcl-2 expressing cells
- • Targets critical residues involved in Beclin 1 BH3 domain binding
Biological Activity Highlights
In Vitro Activity
- • IC₅₀ = 18.2 μM against A172 GBM cells and tumor-initiating stem cells
- • Demonstrates significant anti-proliferative effects in both high and low Bcl-2 expressing cell lines
- • Targets cancer stem-like cells that represent true tumor-initiating populations
- • Shows minimal apoptosis induction while promoting autophagic cell death
In Vivo Efficacy
- • 25 mg/kg dosage effectively reduces tumor growth in animal models
- • Requires lower dosing compared to other BH3 mimetics (e.g., ABT-263 at 75 mg/kg)
- • Demonstrates superior therapeutic window with reduced dosing requirements
- • Maintains efficacy against treatment-resistant GBM stem cell populations
Important Information
- • This molecule is covered by patent protection
- • For laboratory research use only - not for human or veterinary use
- • Requests are reviewed on a case-by-case basis for legitimate research purposes
- • Material transfer agreements (MTA) may be required
- • Response time is typically 3-5 business days
- • For commercial inquiries or licensing opportunities, please contact us separately
